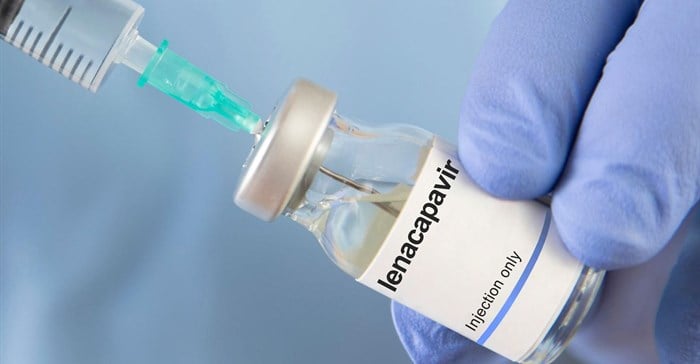
The Ministry of Health has recommended the registration of Lenacapavir, a long-acting antiretroviral medicine, in both tablet and injectable forms, marking a significant step in Kenya’s fight against HIV.
The move comes after a thorough evaluation of the medicine’s quality, safety, and effectiveness by the Pharmacy and Poisons Board, conducted according to Kenyan law and international regulatory standards.
Lenacapavir, available as 300 mg tablets and a 464 mg injectable solution, works by disrupting key stages of the HIV lifecycle, preventing the virus from establishing infection.
Its long-acting formulation is designed to be administered just twice a year, offering a practical alternative to daily oral HIV prevention medicines.
This development is particularly valuable for individuals who struggle with daily pill use due to factors like stigma, adherence difficulties, or pill fatigue.
Kenya is among the first African countries to recommend Lenacapavir for registration, reflecting the nation’s growing capacity to adopt innovative health technologies in line with global public health guidance, including recommendations by the World Health Organization.
Preparations for the initial rollout are underway, with the aim of ensuring the medicine reaches populations at high risk of HIV infection in a timely and equitable manner.
The country has made progress in expanding access to daily oral PrEP across all counties, but long-term adherence challenges remain.
“Long-acting injectable prevention options like Lenacapavir have the potential to address these challenges by reducing how often medicine needs to be taken and broadening access to effective HIV prevention,” Health CS Aden Duale said.
The Ministry of Health reaffirmed its commitment to providing a range of HIV prevention tools, reducing new infections, and advancing the national goal of ending HIV as a public health threat. CS Duale emphasized that “all health products introduced in the country meet the highest standards of safety, quality, and effectiveness.”






























Comments
Sign in with Google to comment, reply, and like comments.
Continue with Google